Different substances in the same sample, for example in a co-polymer, may give the same pyrolysis products, and thus the same peaks in the chromatogram. Is there any way to discriminate between them, or is the situation hopeless? The answer is to look at the formation rates. Even if the resulting pyrolysis product is the same, it is likely that the formation rates are different and then the information can also be used for qualitative information.
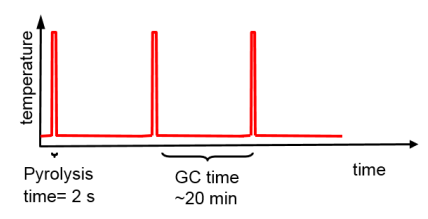
Sequential pyrolysis is a tailor-made method to make the determination of formation rates as straight-forward and accurate as possible. The same piece of sample is pyrolyzed repeatedly to the same temperature, but for so short time that not all of the substance is degraded in one pyrolysis step. The time constant is determined by the total amount of pyrolysis products of all subsequent steps versus the total pyrolysis time. The formation rates are determined in a straight-forward way, removing the inaccuracies due to variations in sample size completely.